The common lipid component of glycosphingolipids, composed of a long-chain amino alcohol (sphingosine) and an amide-linked fatty acid. CelluloseĪ repeating homopolymer of β1-4-linked glucose residues, which is the main constituent of plant cell wall of green plants and many forms of algae and oomycetes and is produced by some bacteria. CAZy databaseĭenoting “Carbohydrate Active enZYmes,” this database describes the families of structurally related catalytic and carbohydrate-binding modules (or functional domains) of enzymes that degrade, modify, or create glycosidic bonds. The domain of a polypeptide that is specifically involved in binding to carbohydrate in lectins, often a highly evolutionarily conserved region of the polypeptide. Includes monosaccharides, oligosaccharides, and polysaccharides, as well as derivatives of these compounds. CarbohydrateĪ generic term used interchangeably in this book with sugar, saccharide, or glycan. Presence of a capsular polysaccharide is often associated with virulence. CapsuleĪ protective extracellular polysaccharide coat surrounding certain bacteria. It is applicable to small quantities of carbohydrates and can interface with a mass spectrometer. Capillary electrophoresis (CE)Īn analytical technology using high voltage across the span of a small-diameter capillary to accomplish separation. Soluble protein chaperone that recognizes N-glycans and mediates quality control of glycoprotein folding in the endoplasmic reticulum. Membrane-bound protein chaperone that mediates quality control of protein folding in the endoplasmic reticulum. C-type lectinsĪ class of Ca ++-dependent lectins recognizable by a characteristic sequence comprising their carbohydrate recognition domain. BiofilmĬommunity of bacteria that adheres to a moist surface (e.g., surface of ponds or teeth). 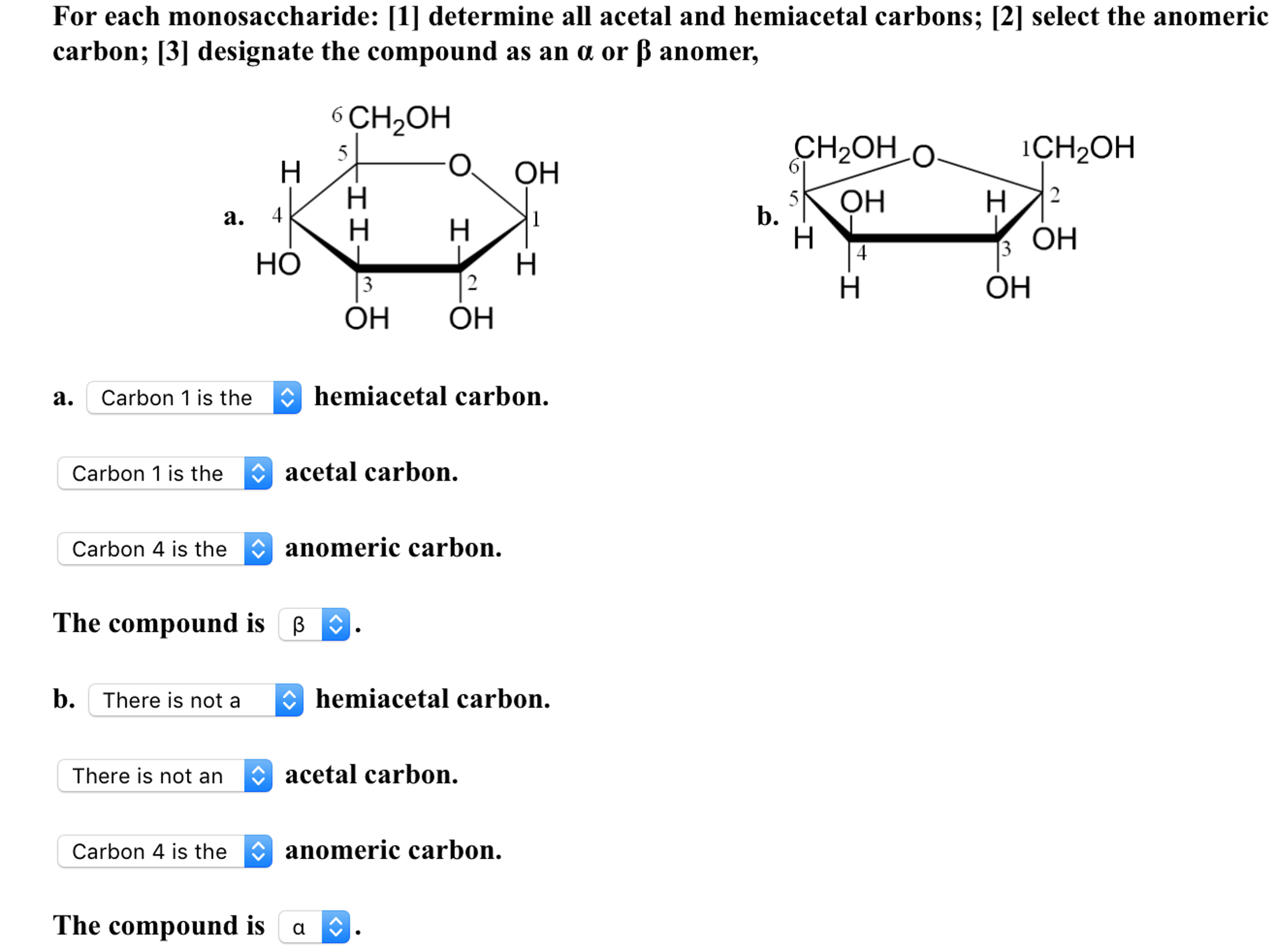
The process is used to cleave O-glycans from Ser or Thr residues. The cleavage of a C-O or C-N bond positioned on the beta carbon with respect to a carbonyl group. Azido sugarĪ monosaccharide to which an azido group has been introduced synthetically. AzideĪ functional group comprising three nitrogen atoms bound in a linear arrangement (N 3). AvidityĪ measure of the combined strength of interaction from the multiple affinities of a multivalent complex. Rapid method for the chemical synthesis of oligo- and polysaccharides on a solid support. 
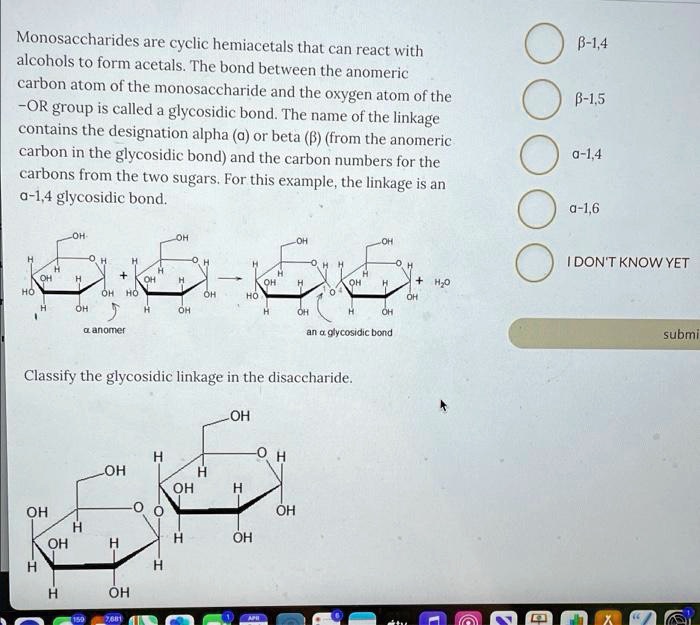
AntennaĪ branch of an oligosaccharide emanating from a “core” structure. Stereoisomers of a monosaccharide that differ only in configuration at the anomeric carbon of the ring structure. The carbon atom of a monosaccharide that bears the hemiacetal functionality (C-1 for most sugars C-2 for sialic acids). Amino sugarĪ monosaccharide in which a hydroxyl group is replaced by an amino group. AldoseĪ monosaccharide with an aldehyde group or potential aldehydic carbonyl group (by definition, this is the C-1 position). Non-carbohydrate portion of a glycoconjugate or glycoside that is glycosidically linked to the glycan through the reducing terminal sugar. The related term hemagglutination denotes the specific case wherein the cells are red blood cells. The clumping of cells in the presence of a protein (e.g., antibody or lectin). 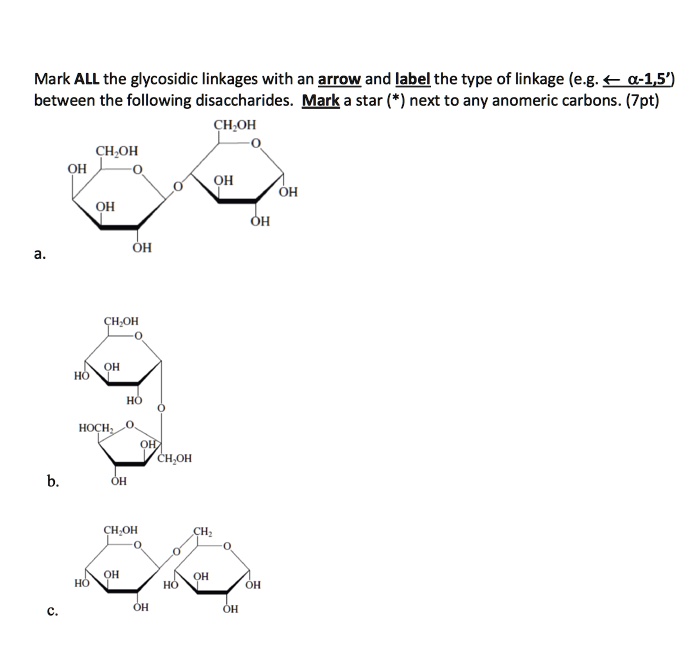
AffinityĪ measure of the strength of interaction between a receptor and its ligand. AdhesinĪ protein on the surface of bacteria, viruses, or parasites that binds to a ligand present on the surface of a host cell. If the hemiacetal is a sugar, the acetal is a glycoside. AcetalĪn organic compound derived from a hemiacetal by reaction with an alcohol. Gene locus comprising three major allelic glycosyltransferases that generate the A, B, and O blood groups.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
